Genetics Discoveries of 2019: In a way, Genetics is a science that studies the most well-kept secrets of life. And in 2019, some secrets the geneticists have uncovered are genuinely astounding.
For instance, it is now clear that cancer mutations can affect multiple genes – because they influence the entire structure of chromosome proteins. We have now found birds with enormous chromosomes – which is both surprising and concerning.
And it is now possible to make our knowledge of genes to be of service to patients, as seen from discussing two promising therapy approaches discussed here: for Duchenne syndrome and for blindness.
Genetic Discoveries in 2019
The following list is just a small presentation of the vast network of genetics research in 2019 that may influence our lives in many unexpected ways.
Getting gene regulation into a cage: a new NET-CAGE method for determination of enhancer structure [Sweden-Japan, September 2019].

Sequencing of the human genome has shown to the scientists that a considerable portion of our DNA does nor code for proteins. At first, these sequences were unfairly called Junk DNA.
Now we know that these DNA fragments are crucial for the regulation of gene activity. There are several types of regulatory sequences. To the scientists at the RIKEN Center for Integrative Medical Science, enhancers were of particular interest:
- Enhancers are sequences that are located at a certain distance from the genes they can influence.
- It is still not clearly understood how enhancers function.
- The researchers have developed a method called NET-CAGE that allows detection and sequencing of enhancers with high sensitivity.
- The method was tested on several types of cancer cell lines.
- The method has allowed finding more than 20000 new enhancers in human cells.
- Unlike promoters that can activate gene transcription in all cell types, enhancers were found to be cell-specific.
This new method would help to understand gene regulation better. Moreover, it is now clear that enhancers play an essential role in cell specialization, as their action is highly specific to cell type.


Sending support for muscle cells: new gene therapy for Duchenne muscular dystrophy developed [Canada, November 2019].
Duchenne muscular dystrophy is a genetic disorder caused by a mutation in the dystrophin gene (DMD). These mutations lead to the loss of dystrophin. This protein provides additional support for the muscle cell membrane, which protects muscle cells from damage.
With the absence of dystrophin due to the switched-off gene, the muscles of the patients degenerate and fail to support the bodily functions.
- The team lead by the medical geneticist Toshifumi Yokota at the University of Alberta decided to improve the existing approach – the use of antisense oligoNucleotides.
- Previously, treatment with antisense oligonucleotides was possible for mutations located out of the frame of the gene.
- The newly developed combination aims at treating the mutations located in the exon 45-55.
- The injection of the oligonucleotide cocktail leads to the shift of the frame during the translation of the defective gene.
- As a result, the dystrophin gene is still produced, though it is shorter compared to normal.
- Such shorter dystrophin proteins are seen in DMD patients with mild symptoms of the disease.
- The treatment was shown to be successful in mice and immortalized human cells from patients with DMD.
If the treatment were proven successful in human subjects, the carriers with more severe mutations subjected to the new procedure would get a significant increase in the quality of life. The researchers predict that the new approach would help treat over 65% of all DMD patients.


A mutation that acts in 3D: cancer cells have a mutation that affects the 3-dimensional structure of chromosomes [Switzerland, January 2019].
Chromosomes have structures called topologically associated domains. These TADs have a high concentration of histone proteins, and the genes located in TADs probably interact with each other.
It is also known that TADs influence gene regulation. The team of researchers from Switzerland has studied the influence of a cancer-related mutation in the gene EZH2 on the TAD function:
- In cancer cells, a mutation in the EZH2 gene leads to methylation in one of the histone proteins.
- The excessive methylation of the histones leads to the overall gene suppression in the cancer cells.
- The methylation driven by the mutated EZH2 protein leads to the preferential silencing of the domains containing multiple tumor suppressors.
- Blocking EZH2 by a specific inhibitor led to the restoration of the normal activity of genes and also healthy chromatin interactions.
This discovery shows that the mutations in cancer may affect entire structures of chromosomes, not only specific genes, and it is essential to consider in cancer therapy.
Currently, there are trials of agents that block the activity of EZH2 mutated protein, which may bring significant benefits to cancer patients.


The history is hidden in our DNA: the researchers have analyzed ancestral DNA to uncover the genetic history of Ancient Rome [USA-Austria-Italy, 2019].
.
Due to the new methodology that allows studying ancient DNA, it has become possible to literally see the history in our genes. An international team that has included researchers from Sapienza University in Rome, Vienna University, Austria, as well as from Stanford, USA, has undertaken a unique study:
- The researchers have taken samples from 29 archaeological sites.
- The sites were dated from Neolithic period, 12, 000 years ago, to the Medieval period.
- There were 127 genomes sequenced in total.
- The first change in the population has occurred at the same time as the ancient people have learned to farm.
- The second change in the human population in Roman territory has occurred around the Iron Age.
- Another significant change has taken place during the Imperial period of Rome, as people started coming to Rome from the Near East and North Africa because of the Roman conquests.
This study not only supports the historical evidence concerning the development and establishment of Rome – but it also shows that analyzing ancient DNA can become another tool that helps us understand what was happening in the past.


The law of Red Queen in action: the scientists have documented the host-parasite co-evolution process [Germany-Switzerland-UK, October 2019].

The famous law of Red Queen states: one needs to run very fast to stay in place. This law is especially true when we look at the process of co-adaptation between hosts and pathogens.
The hosts continuously develop protective measures; the pathogens need to develop ways to overcome them. And all these subtle changes are caused by the changes in the DNA.
To document how the genomes change over time in the host-pathogen pairs, a team of international researchers has decided to create a model host-pathogen system and register the molecular changes over time:
- The researchers have used algae, Chlorella variabilis, as the host and Paramecium bursaria Chlorella virus as the pathogen.
- Both the virus and the algae are not able to reproduce sexually.
- The researchers sampled the population multiple times, observing how both the host and the pathogen adapt to the changes in each other.
- The researchers have documented several parameters:
- Genetic diversity
- Population size
- Evolution of resistance
- Evolution of infectivity
- The host and the pathogen had different dynamics of variability in the population.
- When the host develops resistance towards a new pathogen, there is still considerable diversity in its population, even though the host is protected for a time until the pathogen adapts.
- At the same time, when pathogen becomes more infective due to a mutation, its population has a low level of genetic diversity.
- This pattern may be because the virus has a considerably smaller genome compared to the genome of Chlorella algae, and cannot afford multiple mutations in the population.
It is the first study to link molecular changes and the changes in the population in the course of a prolonged period. It would help significantly in understanding how people change in response to pathogens over time.


The birds with the largest known sex chromosomes discovered [Sweden-UK, December 2019].

A team of researchers has undertaken a comparative genomics study between several bird species namely Eurasian skylark, Horned lark, Raso lark, and Bearded reedling.
The families Panuridae and Alaudidae, to which these species belong, are known for a unique event – the fusion of autosomal chromosomes that leads to the formation of sex chromosomes.
The researchers have analyzed the size and features of the chromosomes in all chosen species and also compared phylogenetic relationships. They have found several interesting facts:
- Eurasian skylark and Raso lark were found to have the largest known sex chromosomes to date, approximately 195.3 Mbp in size.
- The sex chromosomes in these species were the result of a fusion of four autosomal chromosomes over time.
- One of the chromosomes that were part of the fusion, chromosome 3, had an exceptionally high concentration of sex-determining genes.
- The chromosomes that have formed the unusually large chromosomes in larks are similar to sex chromosomes of fish, lizards, frogs, and some mammals.
This discovery is important for two reasons:
- it helps the researchers with understanding how sex chromosomes evolved in different animal groups.
- It points to a potential vulnerability in these particular lark species, as large chromosomes can be especially fragile.


Sometimes the quality is more important than quantity: an algae species was found to have survived despite considerable genome reduction [Korea, October 2019].

It is known that an ancestor of red algae, a highly successful group, has lost approximately 25% of its genome in the course of evolution. A team of researchers from Korea has decided to look into the genetic basis of their success despite such a significant loss.
- The researchers have sequenced the genome of single-cell algae species, Porphyridium purpureum.
- The obtained genome was compared to other known algal genomes to find gene duplications and other features.
- It was found that the chloroplasts and other plastids in these algae have acquired a particularly useful photosynthetic machinery.
- The genome of red algae also contains duplications and variants of genes coding for a vital protein family – phycobilisomes.
- Phycobilisomes act as linker proteins in complex photosynthetic systems inside the cells.
This discovery could help with genetic engineering algae to be used as crops in the future.


New role for the old hero: a new important function of the p53 gene discovered

P53 gene plays a vital role in the cell. It is often called “a guardian ” for it has several known functions:
- Initiation of DNA repair in case of damage.
- Regulation of cell cycle.
- Initiation of cell death if the cell receives irreparable damage.
All those functions are crucial for preventing the cell from becoming cancerous. In cancer development, a mutation of the p53 gene that leads to the loss of these functions is one of the first events that happen to the cell to turn it to the “cancerous ” road.
A collaboration between several labs at the Sloan Kettering Institute has led to a discovery of a previously unknown function of p53:
- The researchers were studying the function of p53 in the cells of the mouse model of pancreatic cancer.
- RNA interference was used to selectively block or restore p53 function in mice.
- It was shown that restoration of p53 activity on pancreatic cancer cells leads to an increase in the level of α-ketoglutarate.
- This is a metabolite that is important for several proteins specializing in modifying chromatine.
- The researchers think that α-ketoglutarate is crucial for tumor-suppressing functions of p53.
There is hope that this discovery would potentially point to a new therapy for cancer.


Sexuality is more complex than previously thought: the specialists have found that homosexuality is not linked with one particular gene [Netherlands-USA, August 2019].

Even now, it is still unclear how homosexuality in humans is determined.
Is it an inherited trait, or can it be influenced by the environment?
This question is very controversial due to the existing attitudes towards the LGBT communities. Despite all the statements to the contrary, as Andrea Ganna and her collaborators have shown, same-sex behavior does have a genetic component:
- The researchers have performed a large-scale genome-wide analysis of the DNA samples taken from USA, UK, and Sweden participants.
- There were 493, 001 participants in total.
- The participants have also filled a questionnaire answering questions about their behavior.
- There were multiple genes associated with same-sex behavior, which points to the polygenic nature of non-heterosexual behavior.
This study has shown that sexuality is of a complex nature and is determined by multiple factors, including genetic ones. There is no single gene that could be switched on-off, which negates all attempts at “gay therapy “, as well as the statement that “being homosexual ” is a choice.


Single gene insertion can restore sight in mice [USA, March 2019]
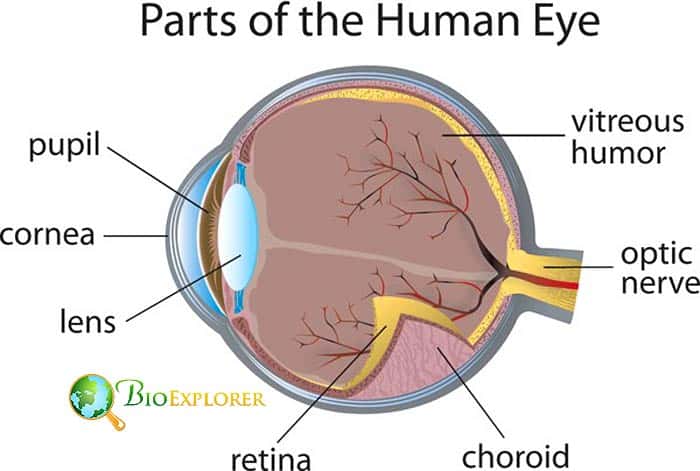
Sometimes, blindness is caused by the loss of cones and rods in the eye due to either an inherited condition or due to the advanced age.
Unfortunately, as there are multiple potential causes for inherited blindness, direct gene therapy is difficult for such patients.
Usually, blind patients are equipped with an electronic eye implant – which is not particularly effective and requires invasive surgery.
The researchers at the University of California – Berkeley have proposed an alternative solution:
- The researchers have used model mice with blindness.
- They have created a viral vector that contains the middle wavelength opsin – a protein that helps the eye detect green light.
- The injection of opsin-carrying vectors into the mice eyes led to the partial restoration of sight in a month after the procedure.
- The mice could determine light patterns on an iPad after undergoing the procedure.
- The use of another sight-related gene, for rhodopsin protein, have not yielded such positive results.
The scientists point out that the protein from the inserted gene can interact with the signaling systems inside the surviving cells, thus partially restoring vision.
A lot of trials and research have to be undertaken before this approach could be used in humans. Still, the idea is highly promising and could help multiple patients.


Genetics helps understand the complex processes in the cell, the behavior of cancer, and the interactions within populations and ecosystems in the course of evolution. All these branches of genetics have yielded exciting news that did not manage to get into the list above.
For instance, it was found that some cancers have a so-called circular DNA that drives their progression. The researchers were able to track not only the co-evolution between pathogens and hosts.
There is new evidence concerning the co-evolution between domesticated crops and humans as well. The new ancient DNA analysis has even helped the specialists in the reconstruction of plague spread in Medieval times.
Do you know what the most astounding news of all is? There could have been more genetic molecules than previously thought. What kind of world would it have been if we had more than one DNA or RNA present in our bodies?
We may not imagine yet – still, with current talent and technology, some must just find out in the future!